Biosimilars in Canada: What Patients Should Know About the Big Switch
Provincial biosimilar switching mandates have sparked patient anxiety across Canada. Here's what the clinical evidence actually shows.
Insights into pharmaceutical innovation, industry trends, and our journey in advancing healthcare through science.

Provincial biosimilar switching mandates have sparked patient anxiety across Canada. Here's what the clinical evidence actually shows.
How Canada's NHP regulations compare to US supplement oversight, what the NPN system requires, and why 'pharmaceutical-grade' is a marketing term.
New research positions TUDCA beyond liver care into neurodegeneration, metabolic disease, and retinal protection. Here's where the science stands.

Generic drugs save Canadians billions, but their prices aren't as low as you'd think. Here's what actually drives the cost.
How gut microbiome composition alters drug efficacy, from cancer immunotherapy to metformin, and why pharmacomicrobiomics may reshape prescribing.
Bioavailability determines how much of a supplement your body actually absorbs. The chemical form, not the milligram count, is what counts.
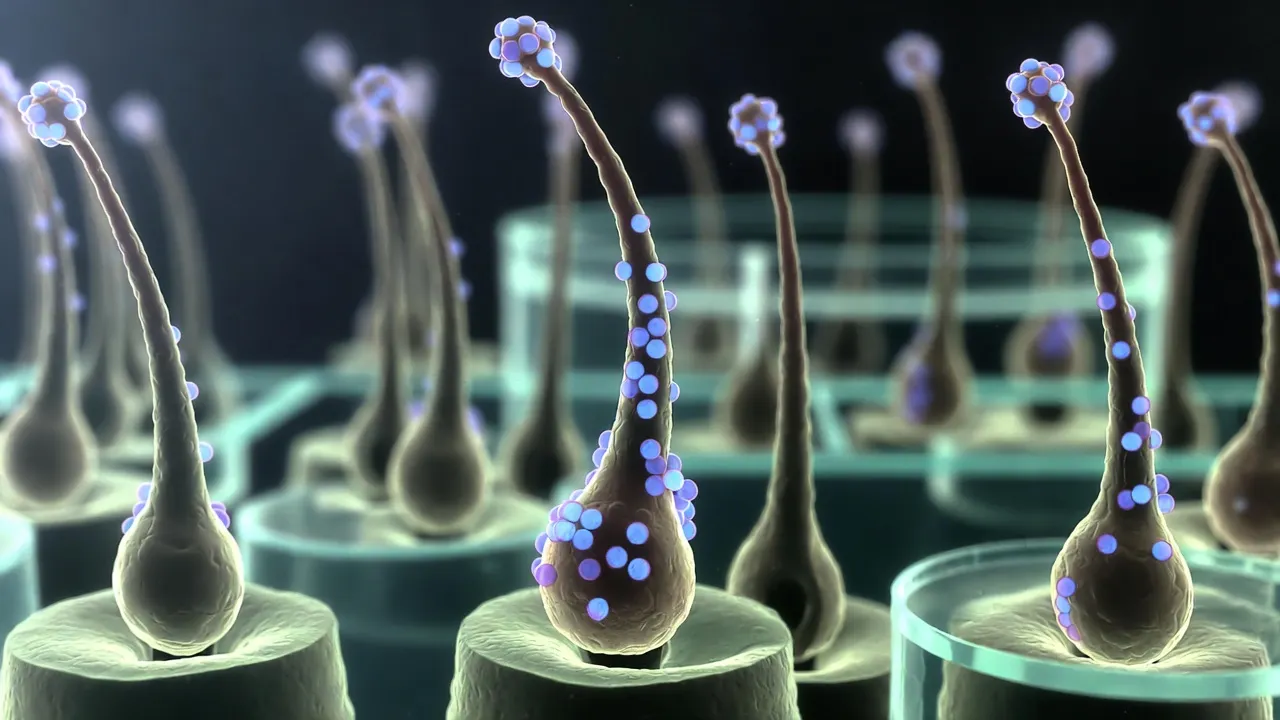
New evidence challenges the long-standing 5% cap on topical minoxidil, as 7% and 10% formulations show promise for hair loss non-responders.

Analysis of opportunities and challenges in Canada's pharmaceutical industry. Explore regulatory frameworks, market dynamics, and growth strategies for pharma companies.

Discover emerging drug delivery technologies revolutionizing pharmaceutical administration. From nanotechnology to transdermal systems, explore innovations improving patient outcomes.